 
This is a key point when teaching Physical chemistry. Make up some practice problems for the future readers. The Aufbau principle is used to predict the electronic configurations of atoms, and accordingly explain the layout of the periodic table, and how the electrons are arranged from low to high energy levels. Learn the definition of the Aufbau principle. You get this by multiplying the three orbitals by 2 electrons per orbital, so 3 multiplied by 2 equals 6. Question: What is the highest number of electrons a 4p subshell can hold?Īnswer: 6! Each 3 p orbital can hold 2 electrons so if they are all filled, the answer is 6. This means there are seven valence electrons.Ħ. A) Aufbau principle - Electrons are added one at a time to the lowest energy orbitals until all electrons have been accounted for. Question: How many valence electrons are there in Iodine?Īnswer: Iodine, z=53, group 17. Question: Demonstrate how elements in a group share similar characteristics by filling in the electron configurations for the Group 18 elements: GroupĪnswer: 1s 2, 2s 22p 6, 3s 23p 6, 3d 104s 24p 6, 4d 105s 25p 6, 4f 145d 106s 26p 6ĥ. Question: What element has the electron configuration 4f 14 5d 10 6s 2 6p 3 ?Ĥ. Question: What element has the electron configuration 4s 2 3d 10 4p 5?ģ. Elements in the same GROUP have similar electron configurations.Ģ. Question, True or False: Elements in the same period have similar electron configurations.Īnswer: False. Groups occupy the vertical rows as opposed to a period which is the horizontal rows in the table of elements. The remaining electron must occupy the orbital of next lowest energy, the 2 s orbital (Figure 8.3.3 or 8.3.4 ). The first two electrons in lithium fill the 1 s orbital and have the same sets of four quantum numbers as the two electrons in helium. After studying the relationship between electron configuration and the period table, it was pointed out by Niels Bohr that electron configurations are similar for elements within the same group in the periodic table. The next atom is the alkali metal lithium with an atomic number of 3. How the periodic table was formed has an intimate correlation with electron configuration. Fully understanding the principles relating to electron configuration will promote a better understanding of how to design them and give us a better understanding of each element in the periodic table. 
Lastly, the Aufbau process describes the process of adding electron configuration to each individualized element in the periodic table. Hund’s rule explains that each orbital in the subshell must be occupied with one single electron first before two electrons can be in the same orbital. The Pauli exclusion rule basically says that at most, 2 electrons are allowed to be in the same orbital. Principles and rules such as the Pauli exclusion principle, Hund’s rule, and the Aufbau process are used to determine how to properly configure electrons. 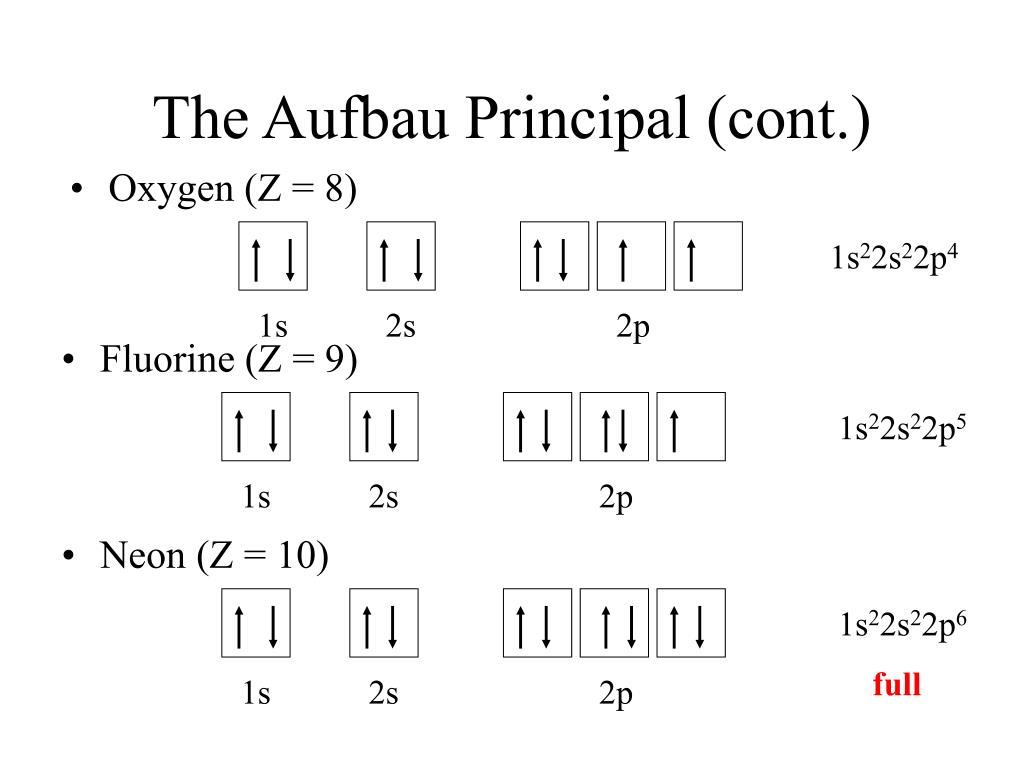
In general when filling up the electron diagram, it is customary to fill the lowest energies first and work your way up to the higher energies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
